HNSCC Cell Research Roadmap
1 Report
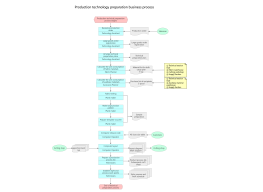
The HNSCC (Head and Neck Squamous Cell Carcinoma) cell research roadmap is a systematic experimental planning framework for elucidating the mechanisms of tumorigenesis, metastasis pathways, and treatment resistance. It aims to reveal the disease progression and screen intervention targets from multiple dimensions, including molecular, cellular, and microenvironmental perspectives. This section details the typical research stages of this roadmap, including five core components: clinical sample collection and primary cell culture, multi-omics characterization, in vitro functional validation, in vivo animal model establishment, and translational validation. In the initial stage, pathologically confirmed HNSCC tissue and paired adjacent normal tissue samples are obtained. Primary cell lines are established using tissue digestion and selective culture methods, and STR identification is performed to ensure lineage traceability. At the multi-omics level, whole-exome sequencing (WES) is used to screen for mutation characteristics (such as TP53, NOTCH1, and PIK3CA), transcriptome sequencing is used to analyze differentially expressed genes and fusion genes, and single-cell RNA sequencing (scRNA-seq) is used to reveal intratumoral heterogeneity and tumor stem cell subsets. In vitro functional experiments include CCK-8 proliferation curves, Transwell migration/invasion, cell scratch healing, and suspension spheroidization assays. Simultaneously, CRISPR-Cas9 or lentiviral overexpression systems are used to validate the functional gain/loss of candidate genes. In vivo models are constructed using subcutaneous xenograft (CDX) or patient-derived xenograft (PDX) models, and tumor growth and lymph node metastasis tendencies are assessed through in vivo imaging and immunohistochemistry. At the translational level, drug sensitivity screening (e.g., cisplatin, 5-FU, cetuximab) combined with organoid drug sensitivity testing can be conducted, and efficacy prediction models can be established based on biomarkers (PD-L1 expression, tumor mutational burden (TMB)). By integrating a temporal sample library and multidimensional validation logic, this roadmap provides a systematic technical roadmap for elucidating the invasion and metastasis mechanisms of HNSCC and developing synergistic targeted therapies.
Related Recommendations
Other works by the author
Outline/Content
See more
Annexin V-FITC/PI double staining flow cytometry
Clinical tissue testing ofHE and IHC
ULBP3
MICA
HNSCC cell
Part 3: To study the antitumor effects of NKT cells and CAR-NKT cells on HNSCC organoids.
Part I: NKG2DL target establishment and CAR-NKT cell construction
Expression of immune-related factors
Histological and molecular level analysis
IFN-γ
Packaging and production of lentivirus
Detection and mechanism analysis of immune factors
Efficacy assessments
Whole exon sequencing (cWES)
HNSCC cell lines
Construction and cell preparation of chimeric antigen receptor
Part 4: Exploring the antitumor activity of NKT cells and CAR-NKT cells in vivo
Preparation and identification of HNSCC organoid model
Direct contact co-culture killing experiment
Indirect co-cultivation experiment:Transwell
ULBP2
Tumor tissue collection
database information mining
surface marker analysis
Preparation of CAR-NKT cells by lentivirus transduction
ULBP1
Cytokine testing: IsoPlexis
V.S.
NKG2DL family is highly expressed in HNSCC
Exploration of immune regulation mechanism
Mouse survival analysis
MICB
Determination of the targeting marker NKG2DL
PI single staining flow cytometry
NKT cells/CAR-NKT cells
Extraction and detection of protein and RNA
Detection of tumor cell effects
Analysis of immune effects and mechanisms
Isolation and transduction of NKT cells
Activation status assessment
Changes in tumor volume in mice
Real-time label-free dynamic cell analysis (RTCA)
Construction of a mouse subcutaneous tumor model
CCK8 testing
Isolation and screening of NKT cells
Collection of PBMC samples in peripheral blood
CAR expression rate
Patient-derived primary tumor cell PDCs
Construction of CAR plasmid vector targeting NKG2DL
High E:T
Part 2: Comparison of the cytotoxic effects of NKT cells and CAR-NKT cells on HNSCC
Gradient verification of different effect-target ratios (E:T)
Co-culture and evaluation of antitumor effect
Flow cytometry was used to detect CAR expression and transduction efficiency
Low E:T
NKT/CAR-NKT cells were co-cultured with PDO
Flow cytometry purity test
Culture supernatant collection
Cell reinfusion therapy and efficacy evaluation
Collect
Collect
Collect
Collect
Collect

0 Comments
Next Page